Steve Jurvetson / Flickr
Steve Jurvetson / FlickrI want to tell you about one of the most beautiful ideas that I know.
It’s a physics experiment, and it’s beautiful because in one elegant stroke, it expands our consciousness, forcing us to realize that objects can behave in ways that are impossible for us to picture (but remarkably, possible for us to calculate). It’s beautiful because it calls into question the bedrock of logic on which we’ve built our understanding of the world. It’s beautiful because it’s deceivingly simple to understand, and yet its consequences are deeply unsettling. And it’s beautiful because I refused to accept it until I ran the experiment for myself, and I distinctly remember watching my worldview shatter as the picture slowly built up on the computer monitor.
This was eleven years ago. I was a college freshman, sitting in a physics lab with all the lights turned out, staring at a blank computer screen, and for reasons that I won’t go into here, listening to a best-of compilation of 80s pop hits.

 Aatish Bhatia
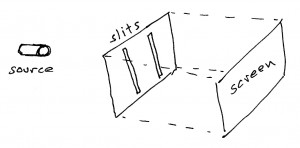
Aatish BhatiaHere’s the setup. On the table in front of me there’s a box with two thin slit-like openings at one end. We’re shooting particles into this box through these slits. I did the experiment with photons, i.e. chunks of light, but others have done it with electrons and, in principle, it could be done with any kind of stuff. It’s even been done with buckyballs, which are soccer ball shaped arrangements of 60 carbon atoms that are positively ginormous compared to electrons. For convenience, I’m going to call the objects in this experiment electrons but think of that word as a stand-in for any kind of stuff that comes in chunks, really.
At the other end of the box is a CCD camera, that takes a snapshot of whatever hits it. Every time a particle makes it to the other side of the box, I see a dot light up at the corresponding point on my computer screen.
Just to be extra careful, we’ve set up the experiment so that there is only one particle inside the box at any given time. Picture, if you like, very tiny baseballs being flung into the box, one at a time. The 80s music plays on, and we sit and wait.
What would you expect to see on the other side of the box? Well, if electrons behaved like waves, you’d expect to see fringes of bright and dark bands, like ripples in a tank of water. That’s because waves can interfere with each other, canceling out when the peak of one wave meets the trough of another, and getting reinforced when the peaks line up.


 Lookang / Wikimedia Commons
Lookang / Wikimedia CommonsBut electrons aren’t waves – they come in chunks. I know this, because I can see them arriving at the screen one at a time, and they strike at a single place, like raindrops falling on dry pavement. And if electrons are chunk-like, then you’d expect to see them piling up behind the slits and nowhere else. In short, you’d expect them to behave like baseballs.

 Aatish Bhatia
Aatish BhatiaAnd indeed, if you do this experiment with only one slit open, they behave just like baseballs, hitting the wall in a single band behind the open slit. A reasonable prediction, then, is that when we run the experiment with both slits open, we should see two bands – one behind each slit.
So what do the electrons do?
Continue reading Hey There Little Electron, Why Won’t You Tell Me Where You Came From?